
Longmont, CO 80501
303-678-1487
http://www.exstrom.com
info[AT]exstrom DOT com
 |
P.O. Box 7651 Longmont, CO 80501 303-678-1487 http://www.exstrom.com info[AT]exstrom DOT com |
A Layman's Introduction to Nuclear Magnetic Resonance
by Stefan and Richard Hollos
Felix Bloch recalling Niels Bohr's description of Nuclear Magnetic Resonance. J. Mattson and M. Simon, The Pioneers of NMR and Magnetic Resonance In Medicine: The Story of MRI (Bar-Ilan University Press, Jericho, 1996). |
Atoms are composed of electrons, protons and neutrons. The protons and neutrons are concentrated in the center of the atom called the nucleus and are surrounded by the electrons. Electrons, protons and neutrons produce their own magnetic fields, behaving like tiny magnets.
Nuclear Magnetic Resonance (NMR) takes advantage of the magnetic properties of the protons and neutrons in the nucleus. The magnetic fields of the protons and neutrons add up to give the nucleus a magnetic field, and sometimes the magnetic fields of the protons and neutrons add up so that the nucleus has no magnetic field. Any nucleus that has a magnetic field can be detected with NMR.
The number of protons in the nucleus of an atom determines which element of the periodic table the atom is. The number of neutrons in a nucleus can vary between a few values depending on the number of protons. Each unique number of neutrons allowed in the nucleus of an element is called an isotope. For example the element hydrogen which contains one proton, may have zero, one, or two neutrons. Thus hydrogen has three isotopes. Most materials contain more than one isotope for each element that is present. Since most elements have at least one isotope with a magnetic nucleus, most elements can theoretically be detected with NMR.
NMR manipulates the magnetic field of the nucleus. The nuclei are placed in a magnetic field generated by permanent magnets which aligns the nuclei similar to the way a compass always aligns itself with the north pole. The nuclei that are aligned in the same direction of the magnetic field will have a lower energy than the nuclei that are aligned opposite to the magnetic field.
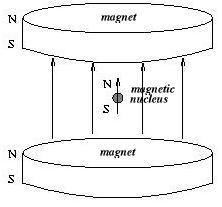 |
 |
|
( lower energy ) |
( higher energy ) |
If the temperature of the material which the nuclei form was at absolute zero, then all the nuclei would be in their low energy state, being aligned with the applied magnetic field. But at any temperature above absolute zero, there will always be some nuclei, although a minority, aligned against the field in their high energy state.
The difference in energy between the nucleus that is aligned with the magnetic field and the same type of nucleus that is aligned against the magnetic field corresponds to a particular energy of an electromagnetic wave. If you impose an electromagnetic wave of that energy on those nuclei, some of the nuclei in the low energy state will absorb the electromagnetic wave and jump up into the higher energy state. This condition is called resonance. The electromagnetic wave with the unique energy for resonance has a corresponding unique frequency.
There are two primary ways that NMR is performed. By the first method, you create an electromagnetic wave and vary its frequency until you detect the wave being absorbed. That method is called "continuous wave NMR". Another method of NMR called "pulse NMR" or "fourier transform NMR" utilizes the fact that the nuclei don't just statically point in the direction of the magnetic field but they spin around the magnetic field. This is because the nucleus has angular momentum. In pulse NMR you send just the right pulse width ( composed of many frequencies of electromagnetic waves ) to cause the nucleus to turn 90 degrees with respect to the external magnetic field, and that causes the nuclei to begin spinning in a plane perpendicular to the external magnetic field. That spinning magnetic field of the nucleus only stays 90 degrees from the external magnetic field for a short time before it starts moving toward being parallel to the magnetic field again. This movement of the magnetic field of the nucleus back toward being parallel with the applied magnetic field, can be detected. The fourier transform of this detected signal gives the frequencies which the nuclei absorbed.
The usefulness of NMR comes from the fact that given a certain strength of externally applied magnetic field, each unique type of nucleus will absorb a particular frequency (equivalent to energy) of electromagnetic wave (due to the uncertainty principle, this won't be a pure frequency but a narrow distribution of frequencies). This allows a positive identification of which nuclei, hence elements, are present in a material. Alternatively, if you already know what type of nucleus is absorbing a particular frequency, then you can determine the magnitude of the magnetic field that the nuclei are immersed in. Thus, one can also use NMR as the basis for a gaussmeter.
But one can extract even more information from NMR. The presence of nearby nuclei changes the energies of the nuclei slightly. This change in energy can be used to determine what nuclei are nearby a given type of nucleus. Therefore, not only can you identify what elements are present in a material, but also in what type of molecule and where in the molecule a detected element can be found. You can also determine how much of an element or molecule is present in a material by the strength of the NMR signal.
In summary, NMR can be used whenever you want to know what some material is made of, and how abundant particular elements and molecules are in the material. NMR is also the most accurate way to measure the strength of a magnetic field.
Copyright 2000 by Exstrom Laboratories LLC